|
Galvanic cells are used as the source of electricity, used in batteries, etc.Įlectrolytic cells are used in electrolysis for purifying metals, etc. In an electrolytic cell, the oxidation reaction occurs at the cathode and the reduction reaction occurs at the anode. In a galvanic cell, the oxidation reaction takes place at anode while the reduction reaction takes place at the cathode. In a galvanic cell, ions discharge at the cathode and consume at the anode.īoth the electrodes of an electrolytic cell discharge the ions. Galvanic cell requires two separate cell containers that form two half cells connected by a salt bridge.Ī single cell container forms the complete electrolytic cell. In case of electrolytic cell, electrical energy flows from external circuit to the cell. In an electrolytic cell, non-spontaneous chemical reactions take place.Ī galvanic cell does not require external voltage source.Īn external voltage source is required in an electrolytic cell.Įlectrical energy flows from cell to the external circuit in a galvanic cell. In case of an electrolytic cell, the anode is positive and cathode is negative.Įlectrons being negatively charged flow from anode to cathode of the galvanic cell.Įlectrons flow from cathode to anode in an electrolytic cell.Ī spontaneous chemical reaction occurs in a galvanic cell to release electrical energy. In an electrolytic cell, electrical energy is converted into chemical energy.įor a galvanic cell, the positive terminal is called cathode, while the negative terminal is called anode. In a galvanic cell, chemical energy is converted into electrical energy. 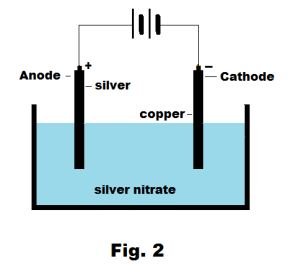
However, there are many differences between a galvanic cell and an electrolytic cell that are listed in the following table − Basis of DifferenceĪ galvanic cell is an electrochemical cell in which chemical energy is transformed into electrical energy.Īn electrolytic cell is an electrochemical cell in which electrical energy is transformed into chemical energy. Difference between Galvanic Cell and Electrolytic Cellīoth galvanic cells and electrolytic cells are types of electrochemical cells. Electrolytic cells are extensively used in electrolysis to decompose the chemical compounds. When a voltage is applied to the electrodes, the ions of the electrolyte are attracted towards an electrode with opposite polarity. Where, the electrolyte is a solution of water and molten slat such as sodium chloride in which ions are dissolved In this way, galvanic cell function to establish an electric current in the external circuit.Īn electrolytic cell is also an electrochemical cell that uses electrical energy from some external source to drive a chemical reaction.Ī typical electrolytic cell has three major parts namely two electrodes (anode and cathode) and an electrolyte. When the circuit of the galvanic cell is closed, the electrons that are released in the oxidation reaction travels through the external circuit to reach another electrode and finally these electrons are used by the reduction reaction. The two electrodes are separated from each other to prevent a direct chemical contact of the oxidation and reduction reactions by creating a potential difference. The galvanic cell consists of two half cells, where each half cell contains an electrode in an electrolyte. What is a Galvanic Cell?Ī galvanic cell is an electrochemical cell in which an electric is produced from spontaneous oxidation-reduction reactions. In this article, we will discuss about galvanic cells and electrolytic cells and how these two types of cells are different from each other.  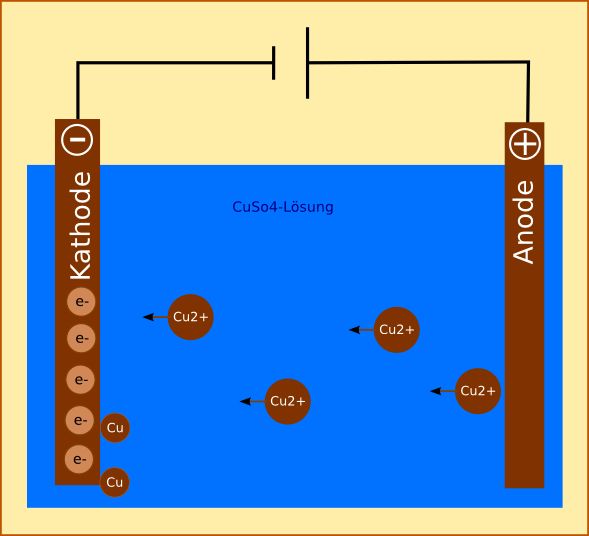
When the cell is connected in a circuit, the chemical reactions take place inside the cell and as a result of these chemical reactions, an electric current flows in the circuit. A cell consists of two electrodes and chemical substances inside it. A cell is a device which can convert chemical energy into electrical energy.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
